 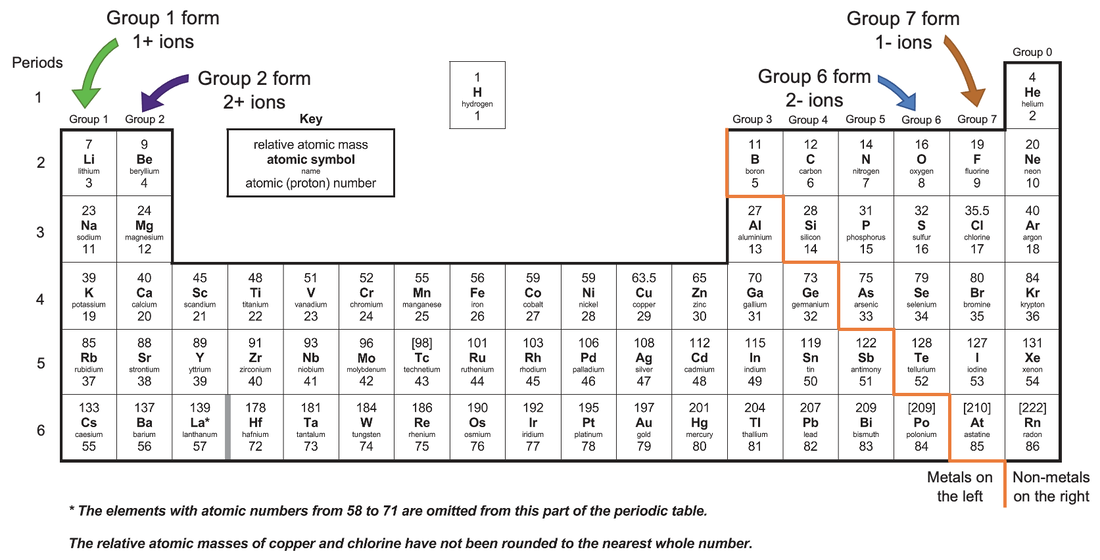
The metal cation is named first, followed by the nonmetal anion as illustrated in Figure 5.7.1 5.7. Lewis, the American chemist who introduced them. A binary ionic compound is a compound composed of a monatomic metal cation and a monatomic nonmetal anion. On the periodic table, atomic radius generally decreases as you move from left to right across a period (due to increasing nuclear charge) and increases as you move down a group (due to the increasing number of electron. For example, it is possible for copper to form both an ion with a +1 charge and to form an ion with a +2 charge. These diagrams are called Lewis electron dot diagrams, or simply Lewis diagrams, after Gilbert N. Atomic and ionic radii are found by measuring the distances between atoms and ions in chemical compounds. Saying an ion has variable charges means that the same element can form ions with different charges. For example, the representation for sodium is as follows:Īnd the representation for chlorine is as follows:įor the above diagrams, it does not matter what sides the dots are placed on in Lewis diagrams as long as each side has a maximum of two dots. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. Second, instead of having a circle around the chemical symbol to represent the electron shell, they have up to eight dots around the symbol each dot represents a valence electron. These diagrams have two advantages over the electron shell diagrams. 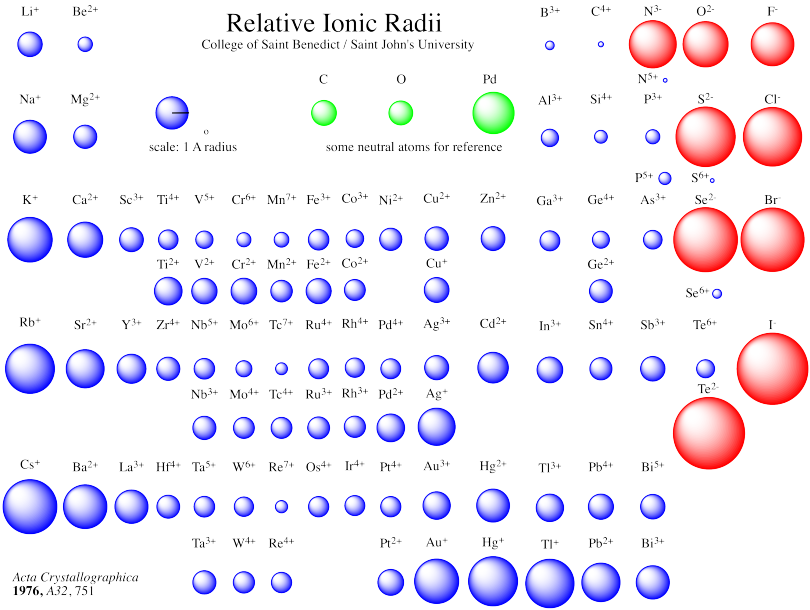
Within a group (family) of elements, atoms form ions of a certain charge.Ĭhemists use simple diagrams to show an atom’s valence electrons and how they transfer. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
